Our Goal
Efficient support for developing safe medical devices
Consulting & Planning
We guide manufacturers through the life cycle stages of medical devices, assuring to meet regulatory requirements
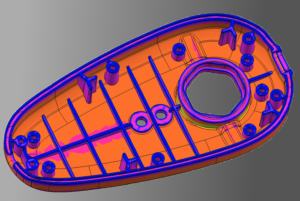
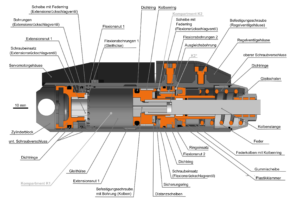
Design & Development
We provide development support in form of simulations, mechanical engineering & design – from concept creation to serial production